February 15, 2021
Journal of Orthopaedic Experience & Innovation: The Clinical and Economic Impact of High Value External Fixation Utilization at a Level II Trauma Center
Use of high value external fixation implant constructs has been a very successful endeavor at our institution providing equivalent clinical outcomes while significantly reducing implant expenditures. Based on this data the use of high value implants has the potential to markedly reduce operative costs as long as quality products are utilized.
INTRODUCTION
In the current health care environment, there has been an increased awareness in the availability and effectiveness of high value or generic orthopaedic implants and their contribution to cost containment (Althausen, Kurnik, et al. 2014; Lybrand and Althausen 2018; McPhillamy et al. 2016). As high value implants continue to be developed for various orthopedic applications it is imperative to demonstrate that these products provide equivalent clinical outcomes to traditional or conventional implants. Without such data, adoption by physicians and cost savings for the healthcare system cannot be realized. The total orthopedic trauma implant market was estimated to be valued at over $5.3 billion in 2016 with an expected 8.3 percent yearly growth. The external fixation market alone in 2016 was estimated to be $716 million with a compound annual growth rate (CAGR) of 5.3% (“U.S. Market for Orthopedic Trauma Devices” 2010). The potential economic impact of high value orthopedic implant use as a cost containment strategy between hospital, surgeon, patients and payors cannot be understated. External fixation devices are often temporary devices placed to allow for soft tissue resolution in comminuted fractures which are removed between 5-10 days after placement when definitive fixation is performed. Most physicians are unaware that these temporary constructs can cost between $5,000 and $10,000 depending on construct type. The purpose of this study is to demonstrate the financial implications of a cost containment program using modern external fixation designs and to examine the clinical outcomes associated with the use of these implants.
MATERIALS AND METHODS
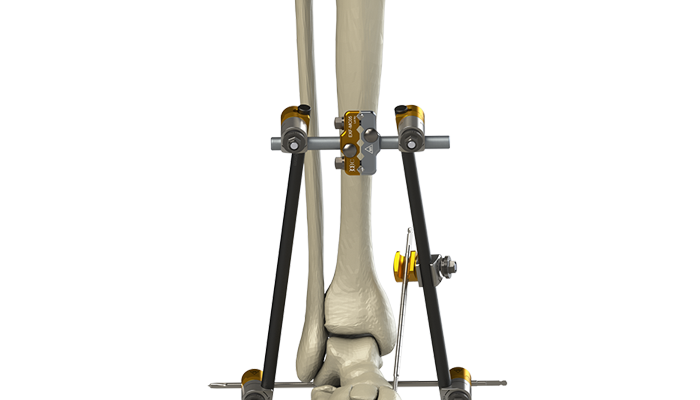
After approval by the institutional review board, 4 of the orthopedic traumatologists at our institution adopted the use of high value external fixation implants (Orthopaedic Implant Company, Reno, NV) beginning in November 2016. Despite a significantly lower cost, these implants were biomechanically tested as equivalent to major conventional implant company products prior to the initiation of the project. Review of our trauma database identified patients with operatively managed tibial plateau and tibial pilon fractures treated with high value implants between November 2016. These patients were then compared to patients treated with conventional implants. Despite slight variation based on surgeon preference, plateau patients were treated with a standard knee spanning construct (Figure 1) consisting of 2 half pins in femur, 2 half pins in tibia, 2 multi-pin clamps, 4 angled posts, 4 pin to bar clamps and 2 carbon bars. Pilon fracture patients were treated with a standard delta frame construct (Figure 2) consisting of 2 half pins in tibia, 1 trans-calcaneal pin, 1 multi-pin clamp, 2 angled posts, 4 pin to bar clamps and 2 carbon bars. All patients underwent similar preoperative, intraoperative and postoperative protocols. External fixation constructs were removed when soft tissues were deemed acceptable for definitive fixation at the time of secondary intervention. Chart review was undertaken to obtain basic demographic variables such as age, sex, smoking and diabetic history. Injury radiographs were reviewed to determine fracture type and OTA Classification. Operative records were reviewed to determine operative time and any adverse intraoperative events. Hospital charts and clinic charts were reviewed to document any postoperative complications. All clinic charts were reviewed by blinded authors to minimize bias. Hospital financial records were appraised to determine implant costs.
Data was analyzed using Microsoft Excel Version 16.32 for Mac. Descriptive statistics were used to describe the data. Two sample T-tests were applied to detect differences of means for continuous variables and chi-square tests were performed to test the differences of proportions of categorical variables between conventional and high value groups. Level of significance was set at p<0.05.
RESULTS
Review of our institutional database identified 62 patients with high value implants which were compared with a consecutive series of 62 patients with similar injuries treated with conventional implants before the arrival of high value implant options. These included both knee and ankle spanning devices.
There were no significant differences in age, sex, presence of diabetes, smoking or fracture type between the high value and conventional groups. (Table 1) No difference in operative time was noted (p= 0.12). No intraoperative complications occurred in either group. (Table 2) Three pin site infections occurred in the conventional group and 4 occurred in the high value group. There was no significant difference in rate of superficial infection between the two groups (p=0.66). There was no occurrence of hardware failure in either group. No complications due to external fixation after conversion to definitive treatment occurred in either group. (Table 3) No significant difference in open fractures (p=0.30) or device construct, ankle vs knee, was observed (p=0.85). (Table 4) OTA classified injury patterns are seen in Table 5.
Overall the hospital realized a 58% reduction in implant costs, an average of $2,200 savings per case and a total savings of $136,400 during the study period.
DISCUSSION
National health expenditures continue to rise, increasing by 4.3% in 2016 to reach $3.3 trillion (“Centers for Medicare & Medicaid Services. National Health Expenditures 2016 Highlights” n.d.). These costs are projected to continue to increase. The total orthopedic trauma implant market was estimated to be valued at over $5.3 billion in 2016 with an expected 8.3 percent yearly growth (“U.S. Market for Orthopedic Trauma Devices” 2010). The external fixation market alone in 2016 was estimated to be $716 million with a compound annual growth rate (CAGR) of 5.3%, extrapolating to $878 million in 2020 (“U.S. Market for Orthopedic Trauma Devices” 2010). The first external fixator was described by Lambote in 1902 and its patents have long since expired (Hernigou and Hernigou 2017). New clamp and pin technology have advanced, making constructs more versatile and easier to apply. Interestingly, unlike other businesses such as the pharmaceutical industry, high value or generic orthopedic implant options have only recently become available. Although there is a tremendous amount of and development invested into new implant designs there is not always a strong correlation with improved clinical outcomes. This cycle continues to escalate the cost of care.
Despite hundreds of articles demonstrating the clinical equivalence of generic medications, only 3 articles were able to be found on generic or value based orthopaedic implants. Waddell et al evaluated generic total hip implants in Canada on 150 patients who were followed over 2 years. The use of generic implants resulted in no increased complication rates and improvement in hip scores (Waddell and Morton 1995). Another article by Althausen et al evaluated the clinical and economic impact of generic 7.3 mm cannulated screws in the treatment of femoral neck fractures and percutaneous sacroiliac fixation. These authors demonstrated a 70% reduction in implant costs with no difference in clinical outcomes (Althausen, Kurnik, et al. 2014). Finally, McPhillamy et al evaluated generic locking plate utilization in a similar study. These authors found a 56% reduction in implant costs with no differences in clinical outcomes (Althausen, Kurnik, et al. 2014). Our study adds to the work of these authors in documenting the economic benefit of high value external fixation constructs with similar clinical outcomes. Our study demonstrates significant cost savings amounting to $136,400 in implant costs alone. This is a 58% reduction in implant costs equating to an average savings of $2,200 per case.
This study adds to growing literature supporting the use of high value implants. However, multiple barriers still exist to value-based implant use. Perhaps most important continues to be the lack of surgeon confidence and general reluctance to adopt a high value implant. In 2016, Walker et al. evaluated surgeon attitudes regarding the use of high value implants in a survey study (Walker and Althausen 2016). While 75% of respondents had an awareness of high value orthopedic implants, only 25% of respondents used them in practice. This is a stark contrast to the 96% of these same respondents that prescribe generic medications regularly. The most common reasons for why respondents had not tried high value implants were satisfaction with current implants, lack of financial incentive to change, and concern regarding track record of high value implants (Walker and Althausen 2016).
As outlined in previous work, multiple barriers to high value implant use exist. Perhaps most important is the lack of surgeon confidence in a high value product. Inaccurate perception of high value implants is propagated by surgeons, implant companies, and hospitals alike.
Although biomechanical equivalence is confirmed preoperatively and all implants approved for use in the United States must meet Food and Drug Administration standards, clinical efficacy must be demonstrated. Because of a general reticence to adopt any type of high value implant, we selected implants that are mechanically sound, conceptually simple, and require no sales representation for implantation. Surgeons must only pick a pin of the appropriate radius and length and pick clamps and bars to create a simple construct. This study evaluates use of very simple devices using techniques with which fracture surgeons have had experience for years. Our data set compares patients treated by traumatologists over a 2-year period, one group with high value implants and one with conventional implants. Equivalence was demonstrated across all operative, postoperative, and radiographic parameters. The only difference was significant cost savings.
Another barrier to change is surgeon’s conflict of interest. At many institutions, orthopaedic traumatologists are paid consultants or have royalty agreements with implant companies. This can make it difficult to effect change. At our institution, none of our trauma surgeons has a consulting agreement or royalty agreement with any of the major branded implant companies. This may play a role in easier adoption of high value implants at our facility. Certainly, conflicts of interest do arise when such relationships exist. This has been a major factor in recent Department of Justice investigations of total hip and knee arthroplasty implant use. For institutions that fear loss of research funding, we advocate that money saved from high value implant use can be appropriated towards research and service line reinvestment. This will free institutions of manufacturer bias or single vendor support.
Another concern raised during high value implant implementation involved the worry that the level of service from existing conventional vendors and sales representatives would decrease or an increase in prices of unique implants and instrumentation would occur. At our institution, the use of high value alternatives over the past 10 years has stimulated better service from conventional companies who wish to preserve their market share. In addition, such dramatic savings have provided the hospital with the ability to more effectively negotiate prices on conventional items such as arthroplasty and arthroscopy products. Matrix pricing has become the norm at our institution with hemiarthroplasty and nail constructs which have resulted in massive savings as well (Althausen, Lapham, and Mead 2016; Althausen, Lu, et al. 2014). As a result, the use of high value alternatives has been successful on many levels.
Cost-effectiveness and comparative effectiveness evaluations are increasing in prevalence in orthopaedic peer reviewed literature. An implant or intervention with equivalent effectiveness that costs 50%–60% less is clearly costeffective and beneficial. Assuming biomechanical equivalence, high value products have a huge potential for cost savings. The credibility and viability of high value implants is directly tied to the capacity of the scientific community to properly test high value implants and ensure that their quality and effectiveness are equivalent to conventional implants. The orthopedic implant market numbers are staggering. If the reduction in costs shown in this study could be applied to other implants, orthopaedic trauma surgeons could have a massive effect on the economics of the healthcare crisis. Given the fact that many of our patients are uninsured or underinsured, we believe that it is our duty to be cost conscious as long as biomechanical equivalency and clinical performance remain equal.
CONCLUSION
The concept of high value implants demonstrates that surgeons do have the capability to positively impact the escalating cost of health care without compromising patient safety or quality of outcomes. As health care resources become more limited innovative cost saving programs will be essential to physicians as a means of preserving patient care standards within an evolving and increasingly complex health care delivery system. Use of high value external fixation constructs has been a very successful endeavor at our institution, demonstrating a 58% savings overall in implant costs compared with conventional implants equating to an average savings of $2,200 per case and total savings of $136,400 over the study period. This has profound implications for the treatment of trauma patients as patents have expired on many other products such as intramedullary nails, locking plates and disposable items such as drill bits. High value implant usage has the potential to markedly reduce operative costs in a manner similar to the high value pharmaceutical industry. As long as quality products are used, patient care is unaffected and cost savings can be realized. A portion of savings from such a change can be reinvested in the hospital trauma program to support OTA/ American Association of Orthopaedic Surgeons position statement guidelines, assist in gain sharing and co-management efforts and positively affect the cost of fracture implants in the future.
Submitted: November 04, 2020 EST, Accepted: November 23, 2020 EST
References:
Althausen, Peter L., C.G. Kurnik, T. Shields, S.F. Anderson, T.P. Gurnea, D. Coll, and M. Lu. 2014. “Clinical and Economic Impact of Using Generic 7.3-Mm Cannulated Screws at a Level II Trauma Center.” American Journal of Orthopedics (Belle Mead, N.J.) 43: 405.
Althausen, Peter L., Joan Lapham, and Lisa Mead. 2016. “Financial Impact of Dual Vendor, Matrix Pricing, and Sole-Source Contracting on Implant Costs.” Journal of Orthopaedic Trauma 30 (December): S37–39. https://doi.org/10.1097/bot.000 0000000000719.
Althausen, Peter L., M. Lu, K.C. Thomas, S.F. Shannon, B.N. Biagi, and E.M. Boyden. 2014. “Implant Standardization for Hemiarthroplasty: Implementation of a Pricing Matrix System at a Level II Community Based Trauma System.” J Arthroplasty Apr;29(4):781-5: 781–85.
“Centers for Medicare & Medicaid Services. National Health Expenditures 2016 Highlights.” n.d. Accessed January 8, 2018. https://www.cms.gov/research-statis tics-data-and-systems/statistics-trends-andreports/n ationalhealthexpenddata/downloads/highlights.
Hernigou, P., and P. Hernigou. 2017. History of External Fixation for Treatment of Fractures. International Orthopaedics. Vol. 41. Berlin/ Heidelberg: Springer.
Lybrand, K.E., MD, and Peter L. Althausen MD, MBA. 2018. “The Role of Value-Based Implants in Orthopedic Trauma.” Orthopedic Clinics of North America, The. United States: Elsevier Inc 49: 437–43.
McPhillamy, Austin, Taylor P. Gurnea, Alastair E. Moody, Christopher G. Kurnik, and Minggen Lu. 2016. “The Clinical and Economic Impact of Generic Locking Plate Utilization at a Level II Trauma Center.” Journal of Orthopaedic Trauma 30 (December): S32–36. https://doi.org/10.1097/bot.00000000000007 21.
“U.S. Market for Orthopedic Trauma Devices.” 2010. Vancouver, BC, Canada: Data Research Inc. https://w ww.idataresearch.net.
Waddell, J.P., and J. Morton. 1995. “Generic Total Hip Arthroplasty.” Clinical Orthopedics and Related Research, 109–16
Walker, Justin A., and Peter L. Althausen. 2016. “Surgeon Attitudes Regarding the Use of Generic Implants: An OTA Survey Study.” Journal of Orthopaedic Trauma 30 (December): S27–31. http s://doi.org/10.1097/bot.0000000000000722.
